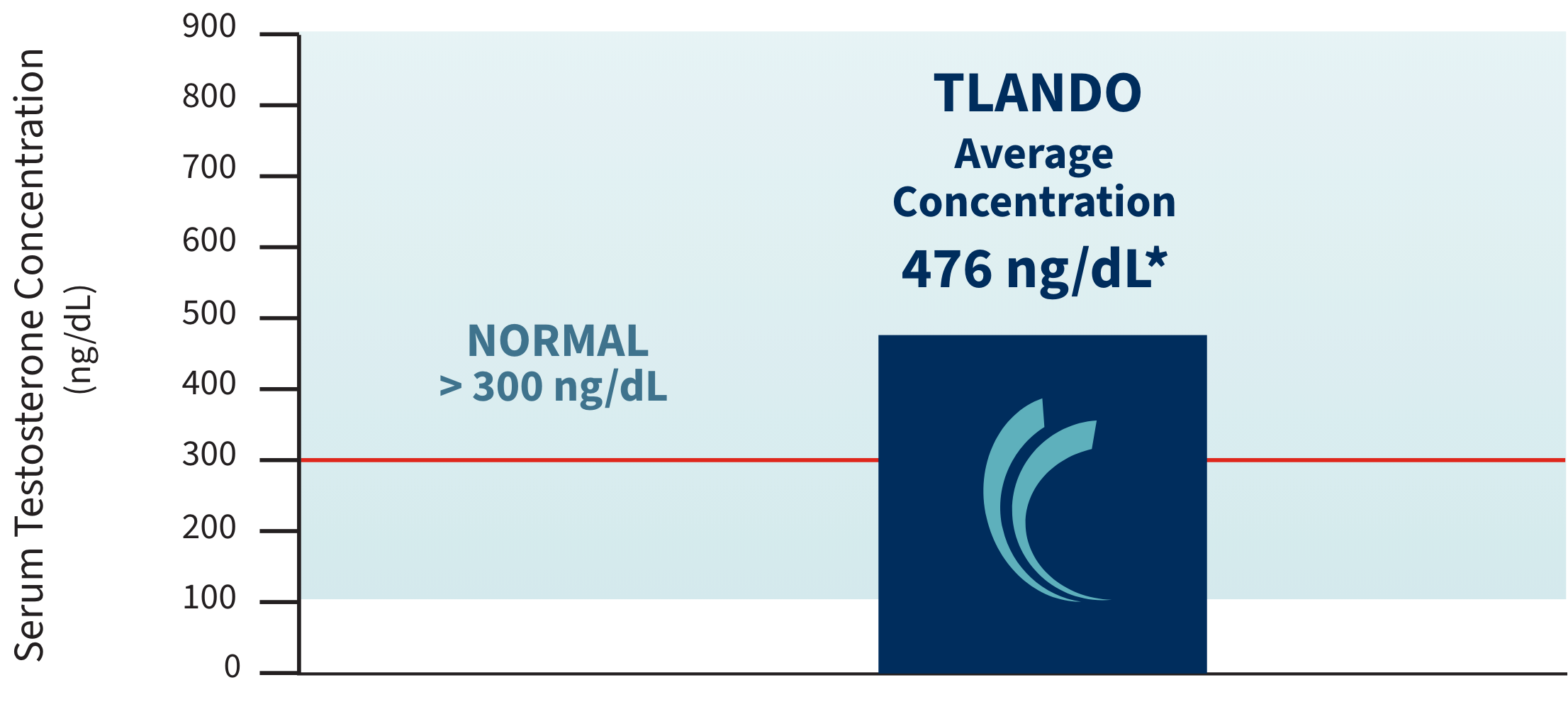
TLANDO INDICATIONS AND USAGE
TLANDO (testosterone undecanoate) is indicated for testosterone replacement therapy in adult males for
conditions associated with a deficiency or absence of endogenous testosterone.
- Primary hypogonadism (congenital or acquired): testicular failure due to conditions such as
cryptorchidism,
bilateral torsion, orchitis, vanishing testis syndrome, orchiectomy, Klinefelter’s syndrome,
chemotherapy, or
toxic damage from alcohol or heavy metals. These men usually have low serum testosterone
concentrations
and gonadotropins (follicle stimulating hormone (FSH), luteinizing hormone (LH)) above the normal
range.
- Hypogonadotropic hypogonadism (congenital or acquired): gonadotropin or luteinizing
hormone-releasing
hormone (LHRH) deficiency or pituitary-hypothalamic injury from tumors, trauma, or radiation. These
men have
low testosterone serum concentrations but have gonadotropins in the normal or low range.
LIMITATIONS OF USE
- Safety and efficacy of TLANDO in males less than 18 years old have not been established.
- Safety and efficacy of TLANDO in men with “age-related hypogonadism” (also referred to as
“late-onset
hypogonadism”) have not been established.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
TLANDO is contraindicated in:
- Patients with carcinoma of the breast or known or suspected carcinoma of the prostate.
- Women who are pregnant. Testosterone can cause virilization of the female fetus when administered to
a
pregnant woman.
- Known hypersensitivity to testosterone undecanoate or any of TLANDO’s ingredients.
WARNINGS AND PRECAUTIONS
Polycythemia: Check hematocrit prior to initiating TLANDO and evaluate every 3 months
during
the first year of treatment, and then every 6 months thereafter while the patient is taking TLANDO. If
hematocrit becomes elevated, stop TLANDO until hematocrit decreases to an acceptable concentration. If
hematocrit increases again after TLANDO is restarted, stop treatment permanently.
Venous Thromboembolism: There have been post marketing reports of venous thromboembolic
events, including deep vein thrombosis (DVT) and pulmonary embolism (PE), in patients using testosterone
replacement products such as TLANDO. Evaluate patients who report symptoms of pain, edema, warmth, and
erythema
in the lower extremity for DVT and those who present with acute shortness of breath for PE. If a venous
thromboembolic event is suspected, discontinue TLANDO and initiate appropriate workup and management.
Worsening of Benign Prostatic Hyperplasia (BPH) and Potential Risk of Prostate
Cancer: Monitor
patients with BPH for worsening signs and symptoms. Evaluate patients for prostate cancer, including
measurement
of prostate specific antigen (PSA), prior to initiating and during treatment with androgens.
Blood Pressure Increases: TLANDO can increase blood pressure which can increase
cardiovascular
risk over time. Monitor BP periodically in men using TLANDO, especially men with hypertension. TLANDO is
not
recommended for use in patients with uncontrolled hypertension.
Abuse of Testosterone and Monitoring of Serum Testosterone Concentrations: Testosterone
has
been subject to abuse, typically at doses higher than recommended for the approved indication and in
combination
with other anabolic androgenic steroids. If abuse is suspected, check serum testosterone concentrations
to
ensure they are within therapeutic range. Counsel patients concerning the serious adverse reactions
associated
with abuse of testosterone and anabolic androgenic steroids.
Not for Use in Women: TLANDO is not indicated for use in women.
Potential for Adverse Effects on Spermatogenesis: Large doses of androgens like TLANDO
can
suppress spermatogenesis. Inform patients of this risk before prescribing TLANDO.
Hepatic Adverse Effects: Prolonged use of high doses of alpha-alkyl androgen has been
associated with serious hepatic adverse effects, however TLANDO is not this type of compound and
therefore is
not known to cause serious hepatic adverse effects. Nonetheless, patients should be instructed to report
any
signs or symptoms of hepatic dysfunction. If these occur, promptly discontinue TLANDO while the cause is
evaluated.
Edema: Edema may occur in patients with or without congestive heart failure and with
preexisting cardiac, renal or hepatic disease. In addition to discontinuing TLANDO, appropriate workup
and
management of edema may be required.
Sleep Apnea: TLANDO may potentiate sleep apnea in some patients, especially those with
risk
factors such as obesity or chronic lung diseases.
Lipid Changes: TLANDO may affect serum lipid profile. Monitor patient lipid
concentrations
periodically; if necessary, adjust dosage of lipid lowering drug(s) or discontinue TLANDO.
Increases in Prolactin: Increases in serum prolactin have been reported in patients
treated
with TLANDO in clinical trials. Monitor serum prolactin levels prior to initiation of TLANDO and 3 to 4
months
after starting TLANDO. Discontinue TLANDO if serum prolactin levels remain elevated.
Other warnings to consider: gynecomastia; hypercalcemia in cancer patients; and decreased
thyroxine-binding
globulin.
ADVERSE REACTIONS
The most commonly reported adverse reactions (≥ 2%) were: increased blood prolactin (6%), hypertension
(5%),
increased hematocrit (4%), upper respiratory tract infection (4%), weight increased (2%), headache (2%),
and
musculoskeletal pain (2%).
DRUG INTERACTIONS
Insulin: TLANDO can cause changes in insulin sensitivity or glycemic control. Androgens
may
decrease blood
glucose requiring a decrease in the dose of anti-diabetic medication.
Oral Anticoagulants: Anticoagulant activity may be seen with androgens. Frequent
monitoring of
international
normalized ratio (INR) and prothrombin time are recommended in patients taking anticoagulants,
especially at
initiation and termination of androgen therapy.
Corticosteroids: Concurrent use of testosterone with corticosteroids may increase fluid
retention and requires
careful monitoring, particularly in patients with cardiac, renal, or hepatic disease.
Medications that May Also Increase Blood Pressure: Concomitant administration of
medications
or drugs known to
increase BP with TLANDO may lead to additional BP increases.
Dosage and Administration
TLANDO is not substitutable with other oral testosterone undecanoate products.
For more information, call 1-844-VERITY-1 (1-844-837-4891)
Please note that this information is not comprehensive. Please click here for full Prescribing
Information.
To report SUSPECTED ADVERSE REACTIONS, contact Verity Pharma at 1-844-837-4891 or FDA at
1-800-FDA-1088 or www.fda.gov/medwatch.

© 2025 Verity Pharmaceuticals, Inc. All rights reserved. TLA-050 September 2025